23rd February 2022
The first thing I want to say here is that there should be nothing in science that is beyond analysis and potential criticism. Because, once this happens, we can find ourselves in a very dangerous situation indeed. A place of unquestioned acceptance of the accepted narrative, with criticism enforced by the authorities.
Unfortunately, I believe this is the place we have reached with COVID19 vaccination. Here is just one example from the UK.
‘GPs have been warned that criticising the Covid vaccine or other pandemic measures via social media could leave them ‘vulnerable’ to GMC* investigation.’1
*GMC = General Medical Council. This is the body that can strike doctors from the medical register so they cannot work as a doctor.
‘Vulnerable to GMC investigation’. What a deliciously creepy phrase that is, dripping with unspoken menace, whilst pretending to be helpful. It sounds like something the Mafia would come up with.
‘I would keep quiet about this, if I were you.’ Baseball bat tapping gently on the floor. ‘No, this is not a threat, think of it as advice from a friend. We don’t like to see anybody making themselves, or their family, vulnerable, and getting seriously injured now, would we?’
It seems that, unless you prostrate yourself before the mighty vaccine, and intone ‘Our vaccine, which art in heaven, hallowed be thy name…’ and suchlike, you will be attacked from all sides … simultaneously. Indeed, to suggest that vaccines are not perfect in every way is the twenty first century’s equivalent of blasphemy.
‘he said Jehovah. Stone him.’
This does make any discussion on vaccines somewhat tricky. To criticize any individual vaccine, indeed any aspect of any individual vaccine, is also to be instantly defined as an anti-vaxxer. Then you will be furiously fact-checked by someone with a fine arts degree, or suchlike, who will decree that you are ‘wrong’.
At which point you will be unceremoniously booted off various internet platforms – amongst other sanctions open to the ‘vulnerable’. This includes, for example, finding yourself struck off the medical register, and unable to earn any money:
‘Hell, we ain’t like that around here. We don’t just string people up, son. First, we have a trial to find ‘em guilty, only then do we string ‘em up. Yeeee Ha!’
Spit … ding!
Yes, it seems you must support the position that all vaccines are equally wonderful, no exceptions. Try this with any other pharmaceutical product. ‘He doesn’t think statins are that great, so he obviously believes that antibiotics are useless.’ Would this sound utterly ridiculous?
But with vaccines… All are the same, all are great, not a problem in sight? I said, NOT! a problem in sight. However, I genuinely believe there are some questions which still have not been answered and simply because of the different types of vaccines that are available, no, not all vaccines are the same.
Just for starters, vaccines come in many different forms. Live, dead, those only containing specific bits of the virus, and suchlike. Now we have the brand new, never used on humans before, messenger RNA (mRNA) vaccines. So no, all vaccines are not alike. Not even remotely.
In addition to the major difference between vaccines, the diseases we vaccinate against vary hugely. Some are viruses, others bacteria, others somewhere in between, TB for example.
Some, like influenza, mutate madly in all directions. Others, such as measles, do not. Some viruses are DNA viruses – which tend to remain unchanged over the years. Others, e.g. influenza, are single strand RNA viruses, and they mutate each year.
Adding to this variety, some of those viruses which mutate very little, also have no other host species to hide in. Smallpox, for example. Which means that the virus was unable to run away and hide in, say, a chicken, or a bat. Others are fully capable of flitting from animal species to animal species. Bird flu and Ebola spring to mind.
Some vaccines just haven’t worked at all. For over thirty years, people have tried to develop an HIV vaccine, and have thus far failed. Early trials on animal coronavirus vaccines also showed some concerning results. Here from the paper ‘Early death after feline infectious peritonitis virus challenge due to recombinant vaccinia virus immunization.’
‘The gene encoding the fusogenic spike protein of the coronavirus causing feline infectious peritonitis was recombined into the genome of vaccinia virus. The recombinant induced spike-protein-specific, in vitro neutralizing antibodies in mice. When kittens were immunized with the recombinant, low titers of neutralizing antibodies were obtained. After challenge with feline infectious peritonitis virus, these animals succumbed earlier than did the control group immunized with wild-type vaccinia virus (early death syndrome).’2
Yet, despite all this massive variety flying in all directions, with some spike protein vaccines found to increase the risk of death (in a few animal studies), attach the word vaccine to any substance, and it suddenly has miraculous properties that transcend all critical thought. Vaccines move in mysterious ways, their wonders to perform.
Yes, of course, some have worked extremely well. The polio vaccine, for example, although I have seen some valid criticisms. Smallpox… I am less certain about. Even though it is held up as the greatest vaccine success story of all. Maybe it was. Smallpox has certainly gone, for which we should be truly thankful. It was a truly terrible disease.
My doubts about the unmatched efficacy of smallpox vaccine simply arise from the fact that diseases come, and diseases go. The plague, for example. This was the scourge of mankind at one time. It tore round and round the world and leaving millions of dead in its wake, over a period of hundreds of years.
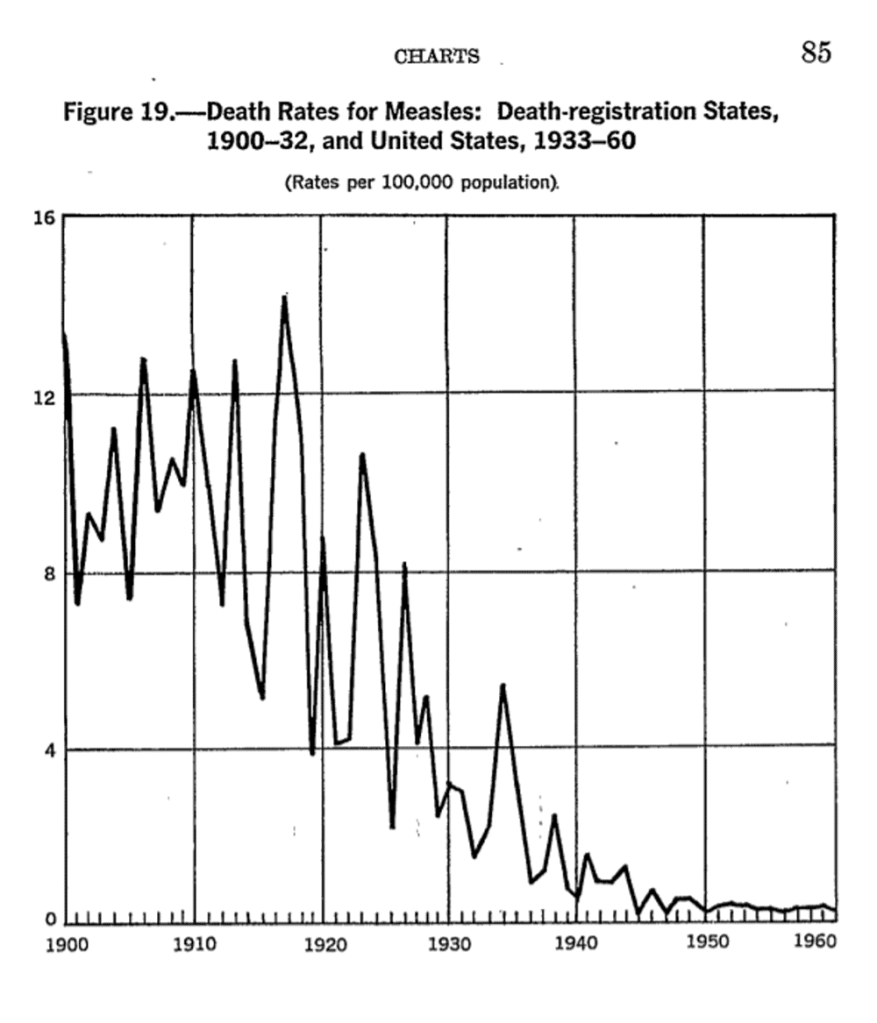
We do not vaccinate against the plague, yet it is virtually unknown today. Cholera killed millions and millions, thousands each year in the UK alone. Now … gone. In the UK at least. This had nothing to do with vaccination either. Measles. There seems little doubt that the measles vaccine is effective. But vaccination cannot explain the fact that measles deaths fell off a cliff and were bumping along the bottom for years and long before we started vaccination programmes.
In the US vaccination did not begin until 1963. So, what happened here? The virus did not mutate, so far as we know. It did not mutate because apparently it cannot. Or, if it did, it would no longer be able to be infective. At least not to humans:
‘While the influenza virus mutates constantly and requires a yearly shot that offers a certain percentage of protection, old reliable measles needs only a two-dose vaccine during childhood for lifelong immunity. A new study publishing May 21 in Cell Reports has an explanation: The surface proteins that the measles virus uses to enter cells are ineffective if they suffer any mutation, meaning that any changes to the virus come at a major cost.’3
So, measles didn’t change, but it did become far less damaging. From around ten deaths per one hundred thousand in the first two decades of the twentieth century, down to much less than one.
Why? What I believe happened with measles is primarily that the ‘terrain’ changed. Nutrition greatly improved. Vitamins, perhaps most importantly vitamin D, were discovered and added to the food supply. Rickets and other manifestation of vitamin D deficiency were rife in the late nineteenth and early twentieth centuries. Virtually gone by 1940.
Of course treatments improved as well, although antibiotics (to treat secondary bacteria pneumonia following measles), did not come into play until the late 1940s, at the earliest.
What we see with measles is simply the fact that infectious diseases have far less impact when they hit a healthy, well nourished person (healthy terrain), than when they hit an impoverished and undernourished child caught in the war in the Yemen, for example.
So, yes, vaccines have played a role in improving human health and wellbeing, but we shouldn’t inflate their impact to the point where they have become the unmatched saviours of humankind. They have certainly not been the only thing that reduced the impact of infectious diseases. They were probably not even the most important thing. ‘Yes … how dare you say this… string up the unbeliever, I know, I know.’
Moving on, and and I think this is even more pertinant to the disucssion that follows. If we cannot accept the possiblility that, at least some vaccines, may have significant adverse effects, if we will not permit anyone to look into this, in any meaningful way. Then we can never improve them. Criticism is good, not bad.
Speaking personally, I do not criticize things that I do not care about. Primarily, because I don’t care if they improve, or not. I only criticize things when I want them to be as good as they possibly can be. It is a character trait of mine to hunt for flaws, and potential problems. Both real and imagined.
Some criticism is, of course, close to bonkers. Suggesting that COVID19 vaccines contain transhuman nanotechnology and microchips of some kind that will become activated by 5G phones … to what end? ‘World domination Mr Bond. Mwahahahahaha etc.’ Quantum dots? Yes, these do exist. But they would be pretty useless at collecting informaiton, and suchlike. Give it fifty years and … maybe.
The problem here is that wild conspiracy theories are simply gathered together with reasonable science-based criticism, to be dismissed as a package of equally mad, unscientific woo-woo tin-foil hat wearing, conspiracy theorist, gibberish.
Which means that, when people (such as me) suggested that COVID19 mRNA vaccination could, potentially, lead to an increased risk of blood clots – this was treated with utter scathing dismissal. I did not understand ‘the science’ apparently. Fact check number one. ‘Oh, look… clots.’
When people questioned the ‘fact’ that the safety phases of the normal clincial trial pathway had been seriously truncated, and that some parts were just non-existent, they were told that they knew nothing of ‘the science’ either.
I looked on the BBC website to find out the ‘official’ party line on vaccine safety information, sanctioned and approved by HM Govt, and SAGE I presume. It was an article entitled ‘How do I know if the vaccine is safe?’ The information rapidly contradicts reality. They say:
- There are different approved types and brands available and all have undergone rigorous testing and safety checks
- Safety trials begin in the lab, with tests and research on cells and animals, before moving on to human studies
- The principle is to start small and only move to the next stage of testing if there are no outstanding safety concerns
The article then looks at fast track approval for vaccines against new variants
- The UK’s drug regulator says new vaccines can be fast tracked for approval if needed.
- No corners will be cut, with safety paramount.
- But lengthy clinical trials with thousands of volunteers will not be needed4
What is wrong here? Well, ‘if the principle is to start small and only move to the next stage of testing if there are no outstanding safety concerns,’ then this principle was not followed. After pre-clinical and animal testing, we move onto trials in humans. Phase I, then II and then III.
Phase I may include as few as twenty people to check that humans don’t simply drop dead on contact with the new agent (it has happened).
Phase II may include a couple of hundred individuals, and usually lasts a few months… a bit more safety, and an attempt to establish the potential size of any health benefit.
Phase III may have up to thirty or forty thousand participants. This phase often lasts for several years.
Well, with the Pfizer Biontech vaccine, the concept of waiting to move to the next stage of testing did not truly occur. Because phase II and III were combined… and the phase III trials have now been, effectively abandoned. They were not supposed to finish until May 2022 at the earliest, and now apparently, they are not going to finish at all. At least not as a double-blind placebo controlled trial.
Yet, we are still informed by the BBC, in all seriousness, that no corners were cut, or will be cut. The fact is that corners were absolutely one hundred per cent cut. Slashed to the bone would perhaps be more accurate. To pretend otherwise is simply to deny reality.
It normally takes around ten years for any drug, or vaccine, to move through the clinical trials process, with each step done in series. COVID19 vaccines took around six months from start to finish, with critical steps done in parallel, and the animal testing was rushed – to say the least. To claim that no corners were cut is nonsense. Nonsense that we are virtually forced to believe?
It is possible/quite likely/probable that vaccine development can be shortened, but please do not tell us that all the normal processes were followed. No-one is that easily fooled.
‘Freedom is the freedom to say that two plus two make four[NK1] . If that is granted, all else follows.’ That freedom disappeared pretty early on in the COVID19 pandemic. I enjoyed the slant that ‘Important quotes explained’ had on the quote from Orwell’s 1984.
By weakening the independence and strength of individuals’ minds and forcing them to live in a constant state of propaganda-induced fear, the Party is able to force its subjects to accept anything it decrees, even if it is entirely illogical.
Of course, it could be that despite the speed with which these vaccines were pushed through nothing important was missed. It is almost certainly true that the standard ten years from start to finish in vaccine and drug development can be compressed, if everyone really wished. Bureaucracy expands to fill the space available.
But in general we are talking about a ten-year process, cut down to six months, or thereabouts. An additional concern is that this happened using mRNA vaccines, which represent a completely new form of technology. One that has never been used on humans before at all, ever.
We are not talking about the sixth drug in a long line of very similar drugs e.g. the statins.
- Lovastatin
- Fluvastatin
- Simvastatin
- Pravastatin
- Atorvastatin
- Cerivastatin
- Rosuvastatin etc.
Statins all do pretty much the exact same thing thing, in exactly the same way. Yet, each one fo them still had to go through the entire laborious clincial trial process. Years and years.
‘Can we not just skip this phase….please?’
‘No.’
‘Please?’
‘No.’
Hold on one moment, just step back, what was that at number six on this list, I hear you say… cerivastatin. You mean you’ve never heard of it. Well, it got through all the pre-clinical trials, then the animal trials. It then sailed through the human Phase II and III trials without a murmur. It was then was launched to wild acclaim. In truth that may be over-egging its real impact, which was a bit more ‘who cares, do we really need another one?’
Here from a 1998 paper: ‘Clinical efficacy and safety of cerivastatin: summary of pivotal phase IIb/III studies.’
‘In conclusion, these studies indicate that cerivastatin is a safe and effective long-term treatment for patients with primary hypercholesterolemia and also suggest that higher doses should be investigated.’ 5
Here from 2001, and an article entitled: ‘Withdrawal of cerivastatin from the world market.’
‘Rhabdomyolysis was 10 times more common with cerivastatin than the other five approved statins. We address three important questions raised by this withdrawal. Should we continue to approve drugs on surrogate efficacy? Are all statins interchangeable? Do the benefits outweigh the risks of statins? We conclude that decisions regarding the use of drugs should be based on direct evidence from long-term clinical outcome trials.’ 6
Yes, as it turns out, cerivastatin caused far more cases of severe muscle breakdown, and death, in a significant number of people. Which meant that it was hoiked from the market.
The moral of this particular story is that, even if you DO do all the clinical studies, fully and completely, one step at a time, over many years, in a widely used class of drug, your particular drug may still be found in the long term, not to be safe. Not even if it is the sixth of its class to launch.
The cerivastatin withdrawal is not an isolated event. You can, if you wish, read this paper ‘Post-marketing withdrawal of 462 medicinal products because of adverse drug reactions: a systematic review of the world literature.’7. So, what happens if you try to compress the entire ten year clinical trial process into around six months, on a completely new type of agent?
… Well then, it may be time to cross your fingers and hope for the best. But please do not insult my intelligence, or the intelligence of anyone else, by trying to tell me that vaccines have undergone: Rigorous testing and safety checks. Compared to what, exactly? Certainly not any other drug or vaccine launched in the last fifty years. ‘We rushed them through, and launched two years before the phase III clinical trials were due to finish.’ would be considerably more accurate.
Two plus two does not equal five, it never has, and it never will. However much you try to browbeat me, and everyone else, into accepting that it does. Indeed, as I write this, the simple fact is that not a single phase III clinical trial has yet ever been completed, on any mRNA COVID19 vaccine, and possibly not ever will be, in truth.
To repeat, this does not mean that mRNA vaccines may not be entirely safe. However, it has become impossible to to claim that we have not seen significant adverse effects from the mRNA vaccines. Effects that were not picked up in any phase of the clincial trials. Here, from the Journal of the American Medical Association in February. One of the most highly cited medical journals in the world:
‘Based on passive surveillance reporting in the US, the risk of myocarditis after receiving mRNA-based COVID-19 vaccines was increased across multiple age and sex strata and was highest after the second vaccination dose in adolescent males and young men.’ 8
I highlighted the first bit here. Namely, the words ‘based on passive surveillance reporting in the US.’ Whilst this adverse effect was not seen, or reported in the clinical trials it was picked up by the passive surveillance reporting system a.k.a. spontaneous reporting systems.
Drug adverse event reporting systems
Frankly, it is surprising that anything at all is ever seen using passive surviellance. In the UK we have the passive/spontaneous reporting system, known as the ‘Yellow Card system.’ In this US (specifically for vaccines) there is ‘VAERS’ (Vaccine Adverse Event Reporting System).
When I use the term ‘spontaneous reporting’, I mean a system whereby someone may (or more likely may not) report an adverse effect to a healthcare professional. They may (or more likely may not) fill in a form, whereupon it goes through to VAERS, who then look at it and can decide whether or not the adverse effect may (or more likely may not) be due to the vaccine. Same basic principle in the UK.
How good are these types of spontaneous reporting system in picking up adverse effects?
Well, as far as I am aware, only one serious attempt has been made to look at how many drug and vaccine-related events were actually reported in the US. Here, from a study by The Agency for Healthcare Research and Quality:
‘Adverse events from drugs and vaccines are common, but under-reported. Although 25% of ambulatory patients experience an adverse drug event, less than 0.3% of all adverse drug events and 1-13% of serious events are reported to the Food and Drug Administration (FDA). Likewise, fewer than 1% of vaccine adverse events are reported.’ 9
Fewer than one per cent of vaccine adverse events are reported. Their words, not mine. Even though, in the US, unlike the UK, there is a legal responsibility to report adverse events – I believe.
When the authors of this report tried to follow up with the CDC and perform further assessment of the system, with testing and evaluation, the doors quietly, but firmly, shut:
‘Unfortunately, there was never an opportunity to perform system performance assessments because the necessary CDC contacts were no longer available and the CDC consultants responsible for receiving data were no longer responsive to our multiple requests to proceed with testing and evaluation.’
This study was done over ten years ago, but nothing about the VAERS system has changed since, as far as I know, or can find out.
In the UK the Yellow Card system may be better, or it may not be. No-one has carried out the sort of detailed analysis that was attempted in the US. However it has been accepted that:
…all spontaneous reporting schemes have a problem with numbers: the MHRA (Medicines and Healthcare products Regulatory Agency) itself says that only 10% of serious reactions and 2 – 4% of all reactions are reported using the Yellow Card Scheme. This means that most iatrogenic* morbidity goes unreported.’ 10
*Iatrogenic means – damage/disease caused by the treatment itself.
Frankly, I see no reason why the Yellow Card system would be any better than VAERS. The barriers to reporting are exactly the same. As the US report states:
‘Barriers to reporting include a lack of clinician awareness, uncertainty about when and what to report, as well as the burdens of reporting: reporting is not part of clinicians’ usual workflow, takes time, and is duplicative.’9
In other words, reporting an adverse event takes an enormous amount of time and effort. You don’t get paid for doing it, you certainly don’t get thanked for it, and you have no idea if anyone paid any attention to it. All made worse if you are not sure if the adverse event was due to the vaccine, or not.
I have filled in yellow cards three times, and several hours of work followed each one. As directed, I searched though patient notes for all previous drugs prescribed, the patient’s medical conditions, a review of the consultations and on, and on. Back and forth from the pharmaceutical company the questions went. Until the will to live was very nearly lost.
If you wanted to devise a system to ensure that adverse effects were under-reported, you could not devise anything better. Yes, doctor, please do report adverse effects to us. The result will be endless hours of work, with no attempt to report back that what you did had the slightest effect, on anything. Thank you for your continued and future co-operation. And yet this, ladies and gentlemen, is the system we have in place to monitor and review all drug and vaccine-related adverse effects.
Which becomes even more worrying because, as mentioned before a couple of times so far, nothing else of much use is going to come out of the clinical trials. With the Pfizer BioNTech trial, crossover occurred in Oct 2020. By crossover I mean the point at which they started giving the vaccine to those in the placebo group as well. End of randomisation, end of useful data. End of … well of anything of any use.
mRNA vaccines and myocarditis
Anyway, getting back to the JAMA study. Even with all the formidable barriers in place to reporting adverse events, JAMA reported an increase in the rate of myocarditis of around thirty-two-fold, as reported via the VAERS system.
I should make it clear that this was the increase seen in the most highly affected population. Males aged eighteen to twenty-four. [Myocarditis = inflammation and damage to heart muscle]. The risk was lower in females, and also in other age groups, although still high. But, to keep things simple, I am going to focus on this, the highest risk group, as far as possible.
The first thing to say is that a thirty-two-fold increase probably does sound enormous. Another way to report this would be, a three thousand one hundred per cent increase, which may sound even more dramatic?
However, myocarditis is not exactly common. In this age group, over a seven-day period, you would expect to see around one and three-quarter cases per million of the population. Multiplying this by thirty-two still only gets you to fifty-six cases per million.
Which is not exactly the end of the world. In addition, most cases may fully recover. Although, having just said this, I have no long-term data to support that statement. The closest condition we have to go on as a comparator, is post-viral infectious myocarditis. And this has a mortality rate of 20% after one year and 50% after five years.11
Which means that myocarditis is certainly not a benign condition of little concern.
Anyway, at this point, you could argue that if around only one in twenty thousand men, in the highest risk population, suffer from myocarditis post-vaccination, then this does not represent a major problem.
It could indeed be worse to allow them to catch COVID19, where the risk of myocarditis is even higher than with vaccination. In reality, we may be protecting them from myocarditis through vaccination. This certainly seems to be the current party line. I might even agree with it…. maybe. So, as is my wont, I looked deeper.
I looked for the highest rate of (reported) post-viral infection myocarditis, in younger people. I believe it can be found here. ‘Risk of Myocarditis from COVID-19 Infection in People Under Age 20: A Population-Based Analysis’ 12
Here, the reported rate was around four-hundred-and-fifty cases per million. On the face of it, this is much higher than the fifty-six cases per million post-vaccination. Approximately ten times as high. But … there are, as always, several very important buts here. There were two key factors that alter the equation.
First, in the JAMA post-vaccine study, the time period for reporting myocarditis was limited to seven days after vaccination. Any case appearing after that was not considered to be anything to do with the vaccine and was thus ‘censored’. In the study above, the time period was far longer. Anything up to ninety days post-infection was counted. A period thirteen times as long.
In addition, although it is difficult to work out exactly what was done from the details provided, the four-hundred-and fifty study only looked at young people who attended outpatients at hospital. These would have been the most severely affected by COVID19, or who had other underlying medical conditions. So, they represent a small proportion, of a small proportion …. of everyone who was actually infected. The vast majority of whom would only have suffered very mild symptoms, or none at all.
In short, we are not remotely comparing like with like here. I find that we very rarely are. We are not only going to vaccinate a small proportion, of a small proportion, of the population who are at high risk of myocarditis. We are going to vaccinate virtually everybody. So, the two populations are completely different.
Leaving that to one side, where else can we look for a comparison between the risk of post-vaccine myocarditis vs post-infection myocarditis. The CDC published this statement.
‘During March 2020–January 2021, patients with COVID-19 had nearly 16 times the risk for myocarditis compared with patients who did not have COVID-19, and risk varied by sex and age.’ 13
Their figure appears to have been entirely derived from a paper published in the British Medical Journal: ‘Risk of clinical sequelae after the acute phase of SARS-CoV-2 infection: retrospective cohort study’ 14. Different age groups were studied here which, again, makes any direct comparison tricky.
This study found a sixteen-fold increased risk, rather than a four hundred and fifty-times risk. A sixteen times risk is around half of the post-vaccination myocarditis risk reported in JAMA, in the eighteen-to twenty-four-year-old group.
Again, though, there were major differences. In the BMJ paper the observation period for inclusion of myocarditis considered to be ‘caused by’ COVID19, was one hundred- and forty-days post infection, not seven days. Twenty times as long for cases to build up.
Equally, after looking at nine million patients records over a year, slightly over two hundred thousand were diagnosed as having had COVID19. Of these, only fourteen thousand had post-infection problems, known as clinical sequelae. In this sub-group, which represents, one point two per-cent of one per-cent of the total, population there were so few cases of myocarditis that they didn’t even appear in the chart published in the main paper. You had to go to supplemental tables and figures 15
To be frank, there are far too many unknowns and uncontrolled variables kicking around here to make any accurate comparisons. However, I do not think it would be unreasonable to suggest that the risk of myocarditis post-vaccination, from these studies, is roughly the same as if you are infected with COVID19.
Once again though, we need to take a further step back. All of our figures here only make sense if all – or the majority of cases of myocarditis – are actually being picked up. What if they are not?
Worst case scenario
SAGE – the UK Governments scientific advisory group for emergencies – have been accused of scaremongering, and only presenting worst case scenarios for COVID19 hospital admissions and deaths. They are not the only ones. This is a worldwide phenomenon.
However, as Sir Patrick Vallance – one of the key members of (SAGE) – has stated, in response to such criticism.
‘It’s not my job to be an optimist’: Sir Patrick Vallance takes swipe at critics accusing scientists of scaremongering over Covid saying ministers need to ‘hear the information whether uncomfortable or encouraging.’ 16
SAGE believe it is their role to highlight the worst possible scenarios, the highest possible death tolls, and such like. So, let us now do the same, and focus on the worst-case scenario regarding mRNA vaccines and myocarditis. Whether ‘uncomfortable or encouraging’.
The worst-case scenario starts like this. If the VAERS system only picks up one per cent of vaccine related adverse effects, this means that we can start by multiplying the JAMA figures by one hundred.
Thus, instead of fifty-six cases per million, the reality is that we could be looking at five thousand six hundred cases per million, post-vaccination. Or very nearly one in two hundred.
If, in this model, we then include the possibility that post-vaccination myocarditis is as damaging as post-viral infection myocarditis, it means that one in four hundred eighteen to twenty-four-year-olds could be dead five years after vaccination.
Do I think that this is likely? I have to say that no, I don’t, really. Although this is where the figures, such as they can be relied upon, inevitably take you. Just to run you through the process a bit more slowly.
- Relying on the VAERS system, JAMA reported a thirty-three-fold increase in myocarditis post COVID19 vaccination. An increase from 1.76, to 56.31 cases per million (in the seven-day period post vaccination)
- It has been established that VAERS may pick up only one per cent of all vaccine related adverse effects
- Therefore, the actual number could be as high as five-thousand six-hundred cases per million ~ 1 in 200.
- Myocarditis (post viral infection) has a mortality rate of 50% over 5 years. So, we need to consider the possibility that post-vaccination myocarditis will carry the same mortality.
- Therefore, the rate of death after five years could be one in four hundred (males aged 18-24)
There are approximately sixteen million men aged between eighteen and twenty-four in the US.
Total number of deaths within five years (men aged eighteen to twenty-four in the US)
16,000,000 ÷ 400 = 40,000
(Divide by five for the UK) = 8,000.
Now, if I were in charge of anything, which I am not, which is probably a good thing, I would hope to have been made aware of these worst-case scenario figures. I would then immediately have begun to do everything I possibly could to verify them.
For starters I would want to know two critical things:
1: Is the VAERS system truly only picking up one per cent of vaccine related adverse effects?
2: Does vaccine related myocarditis lead to the same mortality and morbidity as caused by a viral infection?
If the answer to both of these questions were, yes, then I would have to decide what to do. And that could not possibly, be nothing. At least I would hope not. Yet, nothing appears to be exactly what is currently happening.
As you can tell, I still cling to the concept of ‘first do no harm.’ Today, with COVID19, it seems this this idea has become hopelessly naïve. The current attitude seems to be. ‘We are at war; you must expect casualties’ ‘Also, careless talk costs lives.’ So, my friend, I advise you to keep your ‘vulnerable’ mouth shut, if you know what is good for you.’
Well then, I just hope for everyone’s sake, that these figures are completely wrong. They are, after all, only a model. A worst-case scenario created using the most accurate information available at this time. However, as per the SAGE underlying philosophy, I believe it is important to present the information whether uncomfortable or encouraging.
The thing that most concerns me the most is that we have a worrying signal emerging about the mRNA vaccines. A signal surrounded by a lot of noise, admittedly. Yet, the ‘official’ response continues to be to sweep the entire thing under the carpet. ‘Nothing to see here, move along.’
Postscript
As with regard to the GMC, and the threat of sanctions, as you can see, I am only following their guidance
‘Healthcare professionals must also be open and honest with their colleagues, employers and relevant organisations, and take part in reviews and investigations when requested. They must also be open and honest with their regulators, raising concerns where appropriate. They must support and encourage each other to be open and honest, and not stop someone from raising concerns.’ 17
What do you do if it is the GMC itself that may be stopping someone from raising concerns. Should I report the GMC to the GMC? I imagine they will find themselves innocent of any wrongdoing. Quis custodiet Ipsos custodes?
1: https://www.pulsetoday.co.uk/news/breaking-news/gps-who-criticise-covid-vaccine-on-social-media-vulnerable-to-gmc-investigation/
2: https://europepmc.org/article/MED/2154621
3: https://www.sciencedaily.com/releases/2015/05/150521133628.htm
4: https://www.bbc.co.uk/news/health-55056016
5: https://pubmed.ncbi.nlm.nih.gov/9737644/#:~:text=In%20conclusion%2C%20these%20studies%20indicate,higher%20doses%20should%20be%20investigated.
6: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC59524/
7: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4740994/
8: https://jamanetwork.com/journals/jama/fullarticle/2788346
9: https://digital.ahrq.gov/sites/default/files/docs/publication/r18hs017045-lazarus-final-report-2011.pdf
10: https://wchh.onlinelibrary.wiley.com/doi/pdf/10.1002/psb.1789
11: https://www.ncbi.nlm.nih.gov/books/NBK459259/#:~:text=Immediate%20complications%20of%20myocarditis%20include,and%2050%25%20at%205%20years.
12: https://pubmed.ncbi.nlm.nih.gov/34341797/
13: https://www.cdc.gov/mmwr/volumes/70/wr/mm7035e5.htm
14: https://www.bmj.com/content/373/bmj.n1098
15: https://www.bmj.com/content/bmj/suppl/2021/05/19/bmj.n1098.DC1/daus063716.wt.pdf
16: https://www.dailymail.co.uk/news/article-10341547/Sir-Patrick-Vallance-takes-swipe-critics-accusing-scientists-scaremongering-Covid.html
17: https://www.gmc-uk.org/ethical-guidance/ethical-guidance-for-doctors/candour—openness-and-honesty-when-things-go-wrong/the-professional-duty-of-candour


 The Clot Thickens is available worldwide in Paperback and eBook through Amazon:
The Clot Thickens is available worldwide in Paperback and eBook through Amazon: